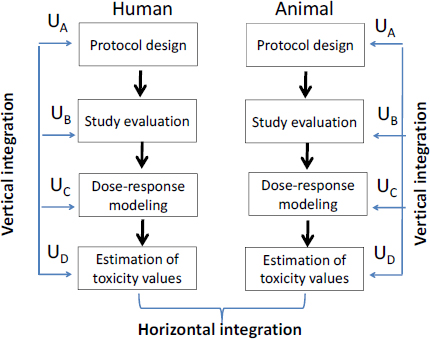
Exposure and toxicity characterization of chemical emissions and chemicals in products: global recommendations and implementation in USEtox | The International Journal of Life Cycle Assessment
Supplemental Module: Human Health Ambient Water Quality Criteria | Water Quality Standards: Regulations and Resources | US EPA

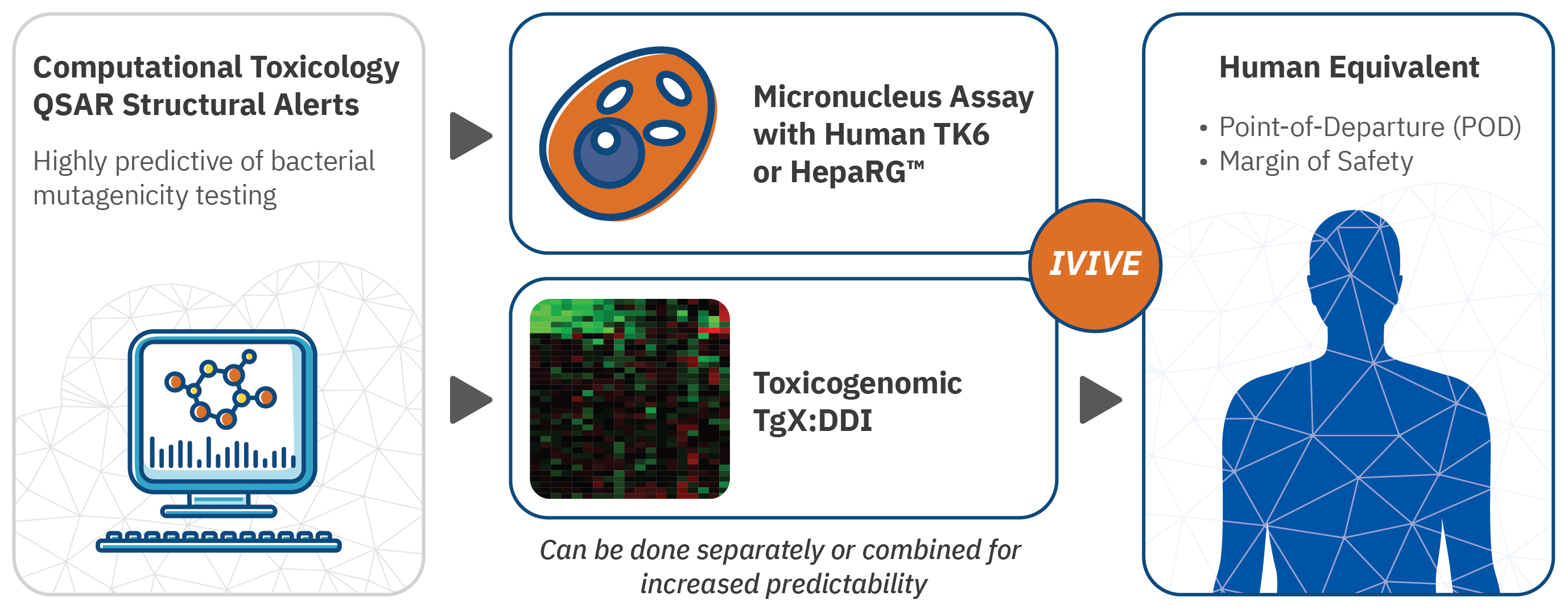
A 10-step framework for use of read-across (RAX) in next generation risk assessment (NGRA) for cosmetics safety assessment - ScienceDirect
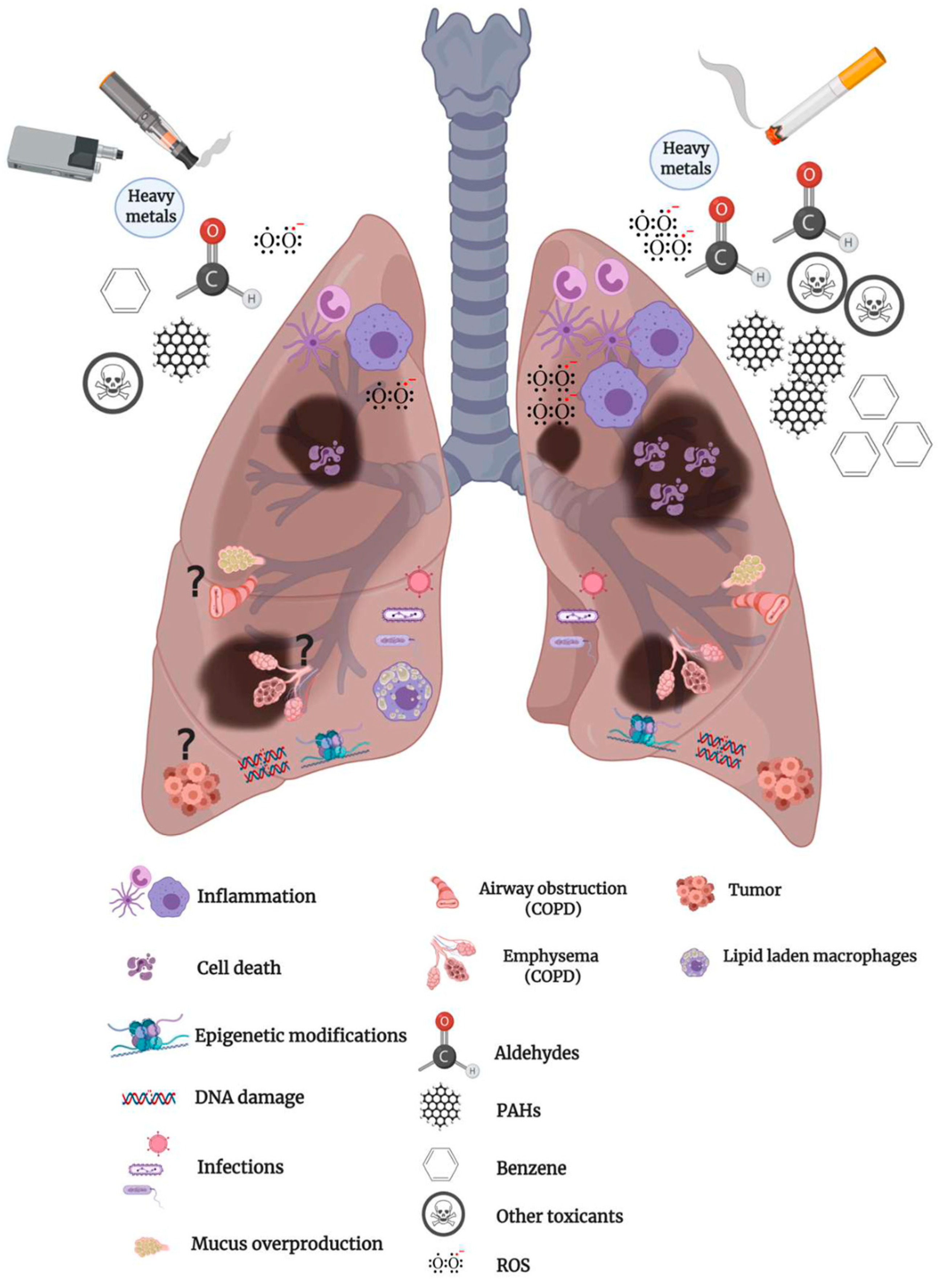
IJMS | Free Full-Text | Inhalation Toxicology of Vaping Products and Implications for Pulmonary Health
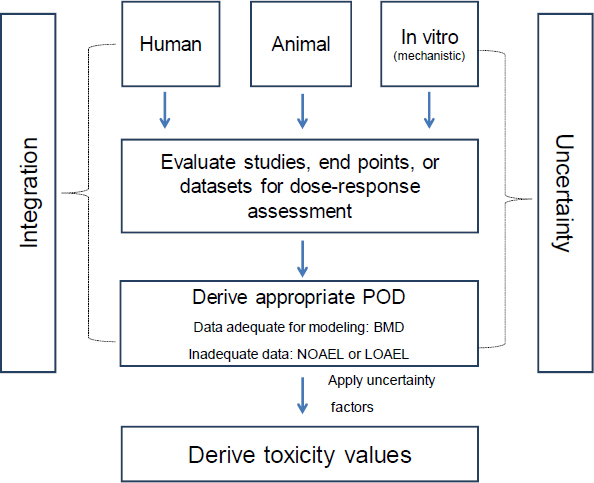
7 Derivation of Toxicity Values | Review of EPA's Integrated Risk Information System (IRIS) Process | The National Academies Press

Silo Pharma Inc. on X: "SP-26, Silo's novel time-released, dosage-controlled formulation of #ketamine, is utilizing @ZyloTherapies' Z- pod technology. The drug revealed positive toxicology results on our path to regulatory approval. Learn more:

Role of toxicokinetics and alternative testing strategies in pyrrolizidine alkaloid toxicity and risk assessment; state-of-the-art and future perspectives - ScienceDirect

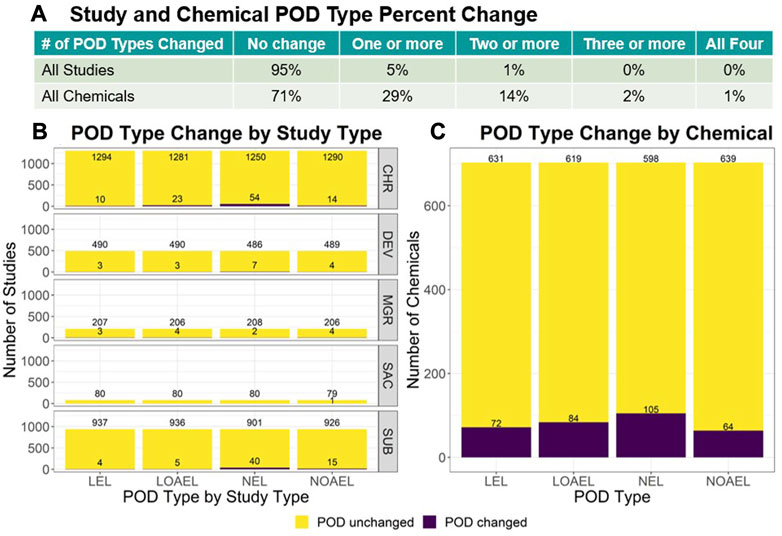
Probabilistic Points of Departure and Reference Doses for Characterizing Human Noncancer and Developmental/Reproductive Effects for 10,145 Chemicals | Environmental Health Perspectives | Vol. 131, No. 3

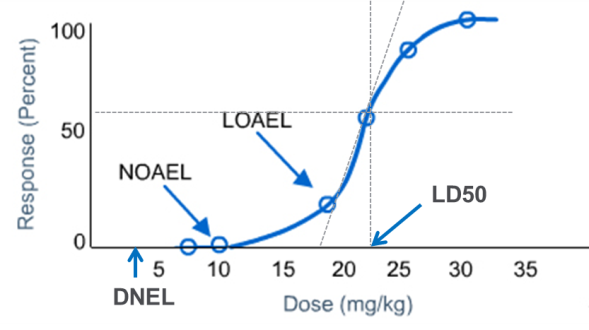
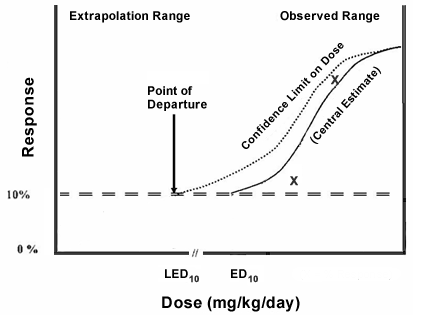
PDF) Derivation of Point of Departure (PoD) Estimates in Genetic Toxicology Studies and Their Potential Applications in Risk Assessment