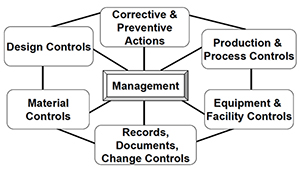
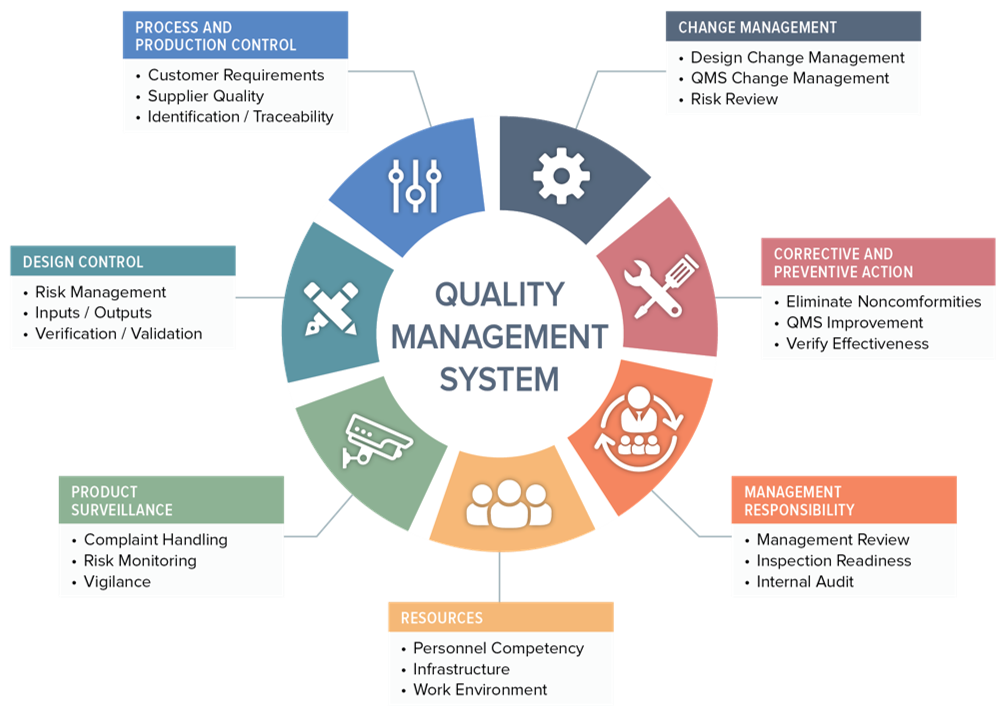
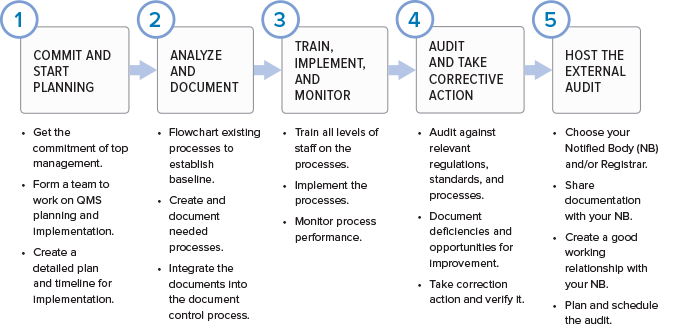
FDA Quality System Regulation for Medical Devices (21 CFR Part 820): A Practitioner's Guide to Management Controls (sections 820.20 Management ... 820.22 Quality Audit, and 820.25 Personnel) : Daugherty, Mr D G: Amazon.fr: Livres

FDA GMP Quality System Regulation: Handling, Storage, Distribution and Installation. : PresentationEZE

The FDA and Worldwide Quality System Requirements Guidebook for Medical Devices, Second Edition | ASQ

FDA Issues Quality Management System Regulation: Final Rule Amending the Quality System Regulation - ICE


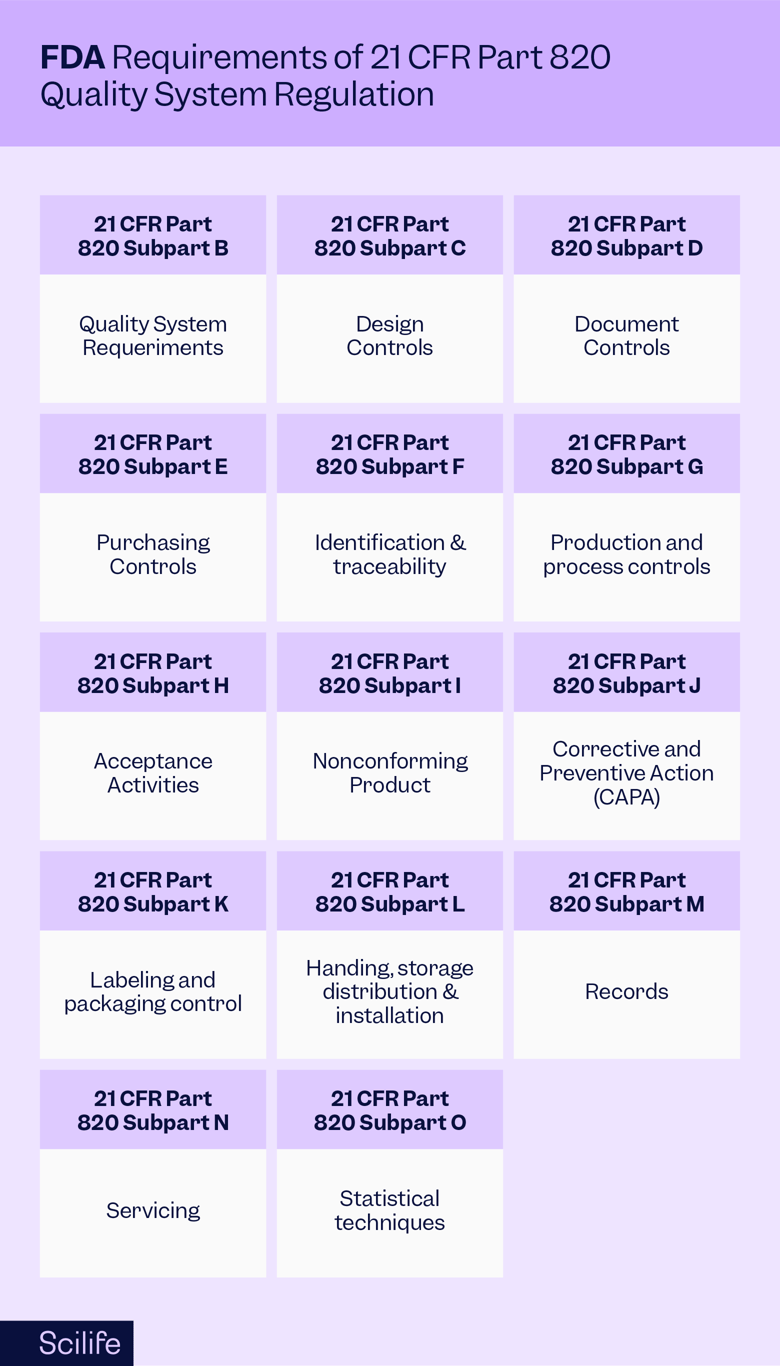

%20Ultimate%20Guide%20to%2021%20CFR%20Part%20820%20%E2%80%94%20FDA%20Quality%20System%20Regulation%20(QSR)%20for%20Medical%20Devices.png?width=250&height=324&name=(cover)%20Ultimate%20Guide%20to%2021%20CFR%20Part%20820%20%E2%80%94%20FDA%20Quality%20System%20Regulation%20(QSR)%20for%20Medical%20Devices.png)